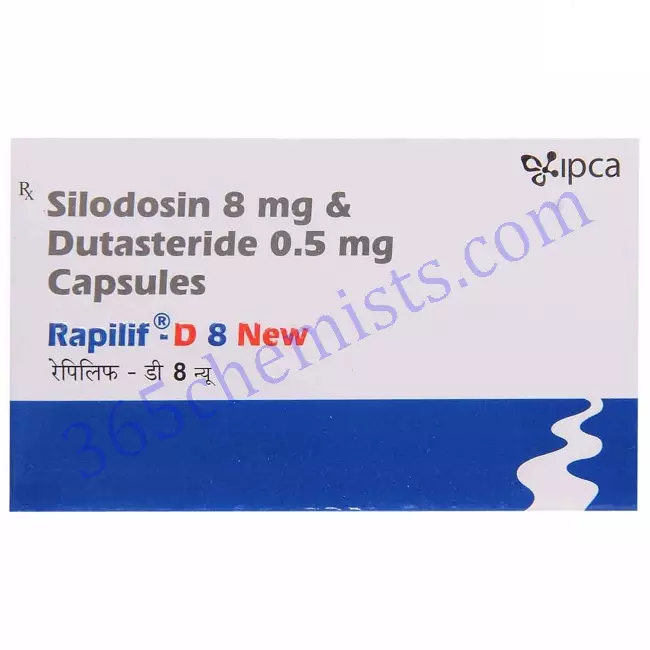
Rapilif-D 8 New Capsule
Rapilif-D 8/0.5 Capsule Combipack is a combination of Silodosin and Dutasteride. It is used to treat the symptoms of benign prostatic hyperplasia (BPH) in men. BPH is a condition that causes enlargement of your prostate gland leading to urinary problems like difficulty in urination, increased frequency, weak urine stream, etc. This medicine works by relaxing the muscles in your urinary bladder, urethra, and prostate. It also blocks the production of a substance (prostate-specific antigen) that causes an enlargement of your prostate.
Rapilif-D 8/0.5 Capsule Combipack shows side effects such as headache, dizziness, diarrhoea, stuffy nose, abnormal ejaculation, and decreased sex drive. These side effects usually subside by themselves. However, consult your doctor if they remain for a long time or become severe.
Rapilif-D 8/0.5 Capsule Combipack should be taken as instructed by your doctor. Your doctor will determine the dose and duration of treatment based on the severity of your condition. Avoid taking more or less than the prescribed dose. For the ease of remembering and better results, take it at the same time every day. Avoid discontinuing this medicine without your doctor’s suggestion, as your symptoms may reappear.
Rapilif-D 8/0.5 Capsule Combipack should be avoided if you are allergic to it or have severe liver/kidney problems. Inform your doctor if you have low blood pressure, any other medical conditions, or taking any other medicines before starting the treatment with this medicine. Rapilif-D 8/0.5 Capsule Combipack is not indicated for use in women and children.
Side effects
Major & minor side effects for Rapilif-D 8/0.5 Capsule Combipack
Chills
Dizziness
Confusion
Headache
Diarrhoea
Stuffy nose
Abnormal ejaculation
Decreased sexual drive
Breast tenderness
Uses of Rapilif-D 8/0.5 Capsule Combipack
What is it prescribed for?
Benign prostatic hyperplasia
Benign prostatic hyperplasia is a condition that causes enlargement of the prostate gland (a small gland in men that secretes fluid to nourish and transport the sperm). As the prostate surrounds the urethra (a tube that carries urine out of your body), an increase in the size of the prostate may lead to symptoms such as difficulty in urination, weak urinary stream, frequent urination, loss of bladder control, etc. Rapilif-D 8/0.5 Capsule Combipack is used in the treatment of BPH.
When not to use?
Allergy
Avoid taking Rapilif-D 8/0.5 Capsule Combipack if you are allergic to it. Serious allergic reactions to this medicine are rare. However, seek immediate medical attention if you notice any symptoms such as skin rash, itching/swelling (especially of the face/tongue/throat), severe dizziness, breathing difficulty, etc.
Severe kidney impairment
Rapilif-D 8/0.5 Capsule Combipack is filtered from your body by the kidneys via urine. Hence, this medicine is not recommended for use if you have severe kidney problems. It may lead to the accumulation of this medicine in your body and increase the risk of side effects.
Severe liver impairment
Rapilif-D 8/0.5 Capsule Combipack is broken down in your liver. Hence, this medicine is not recommended for use if you have severe liver problems as it may cause further liver damage.
Women and children
Rapilif-D 8/0.5 Capsule Combipack is not indicated for use in women and children.
Related Product
Rapilif 4mg Capsule
Rapilif 8mg Capsule
Rapilif D 4 Capsule
Rapilif-D 8 New Capsule
Warnings
Warnings for special population
Pregnancy
Rapilif-D 8/0.5 Capsule Combipack is not intended for use in women.
Breast-feeding
Rapilif-D 8/0.5 Capsule Combipack is not intended for use in women.
General warnings
Cataract surgery
Cataract surgery is an eye surgery performed to replace the lens in your eyes with an artificial lens to correct the blurring of vision. Rapilif-D 8/0.5 Capsule Combipack may lead to complications during cataract surgery. Hence, if you are taking this medicine, inform your doctor before undergoing surgery.
Low blood pressure
Rapilif-D 8/0.5 Capsule Combipack may cause dilation of your blood vessels and lead to a decrease in blood pressure. Hence, this medicine should be used with caution if you have low blood pressure.
Blood donation
It is not recommended to donate blood until 6 months have passed after taking the last dose of Rapilif-D 8/0.5 Capsule Combipack. This measure is taken to prevent the transfusion of blood to pregnant women.
Other medicines
Inform your doctor if you are taking any other medicines as they may interact with Rapilif-D 8/0.5 Capsule Combipack and cause undesired effects.
Use in women and children
Rapilif-D 8/0.5 Capsule Combipack is not indicated for use in women and children.
Driving or operating machines
Rapilif-D 8/0.5 Capsule Combipack may cause a decrease in blood pressure leading to symptoms like dizziness. Avoid performing activities that require high mental alertness like driving vehicles or operating machines if you experience any symptoms during treatment with this medicine.
Dosage
Missed Dose
If you miss a dose of Rapilif-D 8/0.5 Capsule Combipack, take it as soon as you remember. If it is time for your next dose, skip the missed dose. Do not double your dose to make up for the missed dose.
Overdose
Never take more than the prescribed dose of Rapilif-D 8/0.5 Capsule Combipack. Seek emergency medical treatment in case of an overdose.
Interactions
All drugs interact differently for person to person. You should check all the possible interactions with your doctor before starting any medicine.
Interaction with Alcohol
Description
N/A
Instructions
Avoid consumption of alcohol while taking Rapilif-D 8/0.5 Capsule Combipack as it may increase the risk of dizziness, headache, lightheadedness, etc.
Interaction with Medicine
Amitriptyline
Ciprofloxacin
Alprazolam
Nefazodone
Clotrimazole
Erythromycin
Itraconazole
Baclofen
Diltiazem
Chlordiazepoxide
Nelfinavir
Conivaptan
Disease interactions
Kidney Disease
Rapilif-D 8/0.5 Capsule Combipack should be used with caution if you have mild to moderate kidney problems as it may increase the risk of side effects. Your doctor may suggest tests to periodically monitor your kidney function and recommend dose adjustments based on your clinical condition.
Liver Disease
Rapilif-D 8/0.5 Capsule Combipack should be used with caution if you have mild to moderate liver problems as this medicine may cause further liver damage. Your doctor may suggest tests to periodically monitor your liver function while taking this medicine.
Prostate cancer
Cancer of the prostate gland has similar symptoms to Benign Prostatic Hyperplasia. Hence, it is important to know if you have prostate cancer before starting the treatment. Your doctor may suggest tests to rule out the presence of prostate cancer before starting the treatment with Rapilif-D 8/0.5 Capsule Combipack.
Food interactions
Information not available.
Lab interactions
Information not available.
This is not an exhaustive list of possible drug interactions. You should consult your doctor about all the possible interactions of the drugs you’re taking.
General Instructions
Take Rapilif-D 8/0.5 Capsule Combipack as instructed by your doctor. Take this medicine for the recommended duration.
Avoid taking more than the prescribed dose as it may increase the risk of side effects.
Consult your doctor if your symptoms do not improve or become worse.
Avoid blood donation until 6 months of taking the last dose of Rapilif-D 8/0.5 Capsule Combipack to prevent the transfusion of blood to pregnant women.