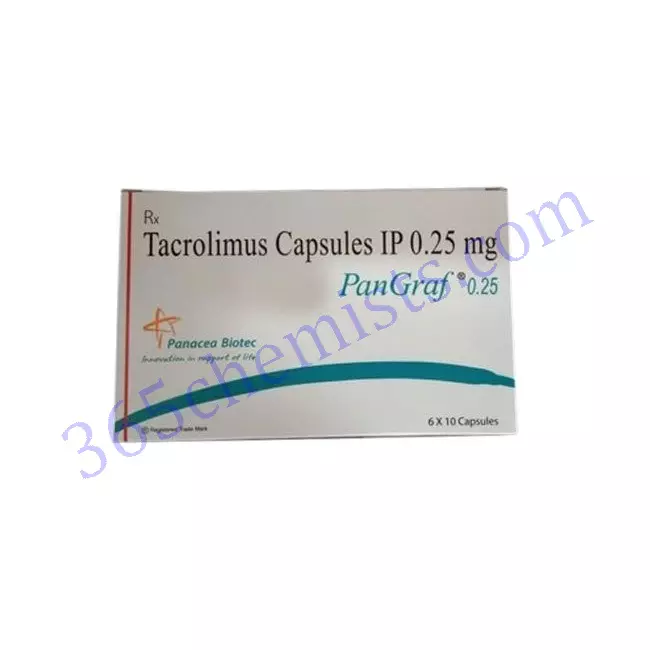
Pangraf 0.25mg Capsule
Pangraf 0.25mg Capsule is a medication that is commonly prescribed as an immunosuppressive agent in patients who have undergone organ transplantation. The active ingredient in this medication is tacrolimus, and the medication is in the form of a capsule. This medication is primarily employed for the purpose of preventing organ rejection. It is classified as a member of a class of drugs known as calcineurin inhibitors. Pangraf 0.25mg Capsule is available in the form of a capsule, and it is typically prescribed in conjunction with other immunosuppressant drugs. In the following paragraphs, we will provide a comprehensive analysis of the Pangraf 0.25mg Capsule, discussing its mode of action, indications, dosage, potential adverse effects, and precautions.
Mechanism of Action
The active ingredient in Pangraf 0.25mg Capsule is tacrolimus, and it works by inhibiting the enzyme calcineurin. Calcineurin is involved in the activation of T-lymphocytes, which are a type of white blood cell that plays an important part in the immune response. Tacrolimus works to reduce the production of cytokines and other immune system molecules by inhibiting calcineurin. These molecules are responsible for the activation and proliferation of T lymphocytes. In organ transplant recipients, this immunosuppressive action helps to prevent the rejection of transplanted organs.
Indications
Pangraf 0.25mg Patients who have undergone organ transplantation, specifically those who have received a kidney, liver, or heart transplant, are the primary target population for the preventative use of capsule. In order to achieve the highest level of immunosuppression possible, it is typically administered in conjunction with a number of other immunosuppressive drugs, including mycophenolate mofetil and corticosteroids. Combination therapy makes it possible to take lower doses of individual medications, which in turn reduces the likelihood of experiencing adverse effects while still providing sufficient immunosuppression.
Related Product
Pangraf 0.25mg Capsule
Pangraf 0.5mg Capsule
Pangraf 1mg Capsule
Dosage and Administration
It is possible for the recommended dosage of Pangraf 0.25mg Capsule to change depending on variables such as the kind of organ that is being transplanted, the patient’s weight, and the particular treatment plan that has been prescribed by the medical professional. It is extremely important to take the medication exactly as directed by the healthcare professional, including paying close attention to the dosage and how it should be administered. Oral administration is the most common method for taking Pangraf 0.25mg Capsule. This medication should be taken on an empty stomach, either one hour before or two to three hours after a meal. It is important to take the capsule as directed and to do so with a full glass of water. It is best to avoid forgetting to take a dose, but if this does happen, the missed dose should be taken as soon as it is remembered, unless it is getting close to the time of the next dose that is scheduled.
Side Effects
Pangraf 0.25mg Capsule has the potential to cause side effects, just like any other medication. Tremors, headaches, diarrhoea, nausea, high blood pressure, and increased susceptibility to infections are some of the most frequently reported adverse effects of this medication. These unwanted effects are typically manageable and have a tendency to become less severe over time. On the other hand, it is essential to make a prompt report to the healthcare provider of any side effects that continue or become severe. In addition, it is possible that using Pangraf 0.25mg Capsule for an extended period of time will increase the risk of developing certain complications, such as kidney damage and diabetes. It is recommended to perform routine checks on both the kidneys’ functions and the glucose levels in the blood.
Precautions
Before beginning treatment with Pangraf 0.25mg Capsule, healthcare providers should evaluate the patient’s medical history and consider the potential risks and benefits that may be associated with the medication. Patients who have a history of hypersensitivity to tacrolimus or any of the components that make up Pangraf 0.25mg Capsule should not take this medication. It is essential to provide the healthcare provider with accurate information regarding any preexisting medical conditions, such as liver or kidney disease, as adjustments to the prescribed dosage may be required. Patients taking tacrolimus should take precautions to prevent sunburn and skin cancer by avoiding exposure to sunlight and other forms of ultraviolet radiation.
Conclusion
Pangraf 0.25mg Capsule (Tacrolimus 0.25) is an immunosuppressive medication that is commonly prescribed to transplant recipients in order to reduce or eliminate the risk of organ rejection. Pangraf 0.25mg Capsule is an important component in the process of preserving the viability of transplanted organs because it works to suppress the immune response. It is common practise to prescribe it in conjunction with other immunosuppressive drugs, and it needs to be monitored on a consistent basis in order to ensure the best possible therapeutic outcomes and reduce the risk of adverse reactions. Pangraf 0.25mg Capsule has significantly increased the success rates of organ transplantation, and it continues to be an essential component of post-transplant care.