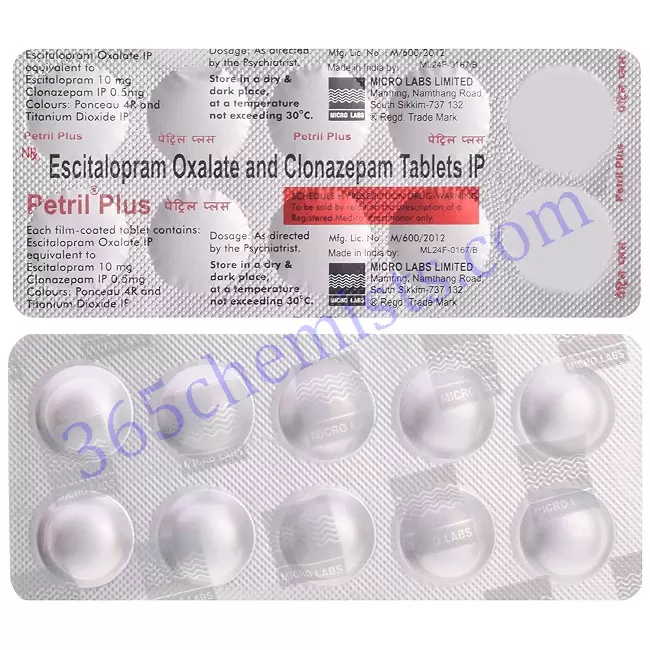
Petril Plus Tablet
Petril Plus (10/0.5 mg) Tablet is a combination medicine used for the treatment of patients suffering from both depression and anxiety disorders. This medicine is not recommended for use in patients below 18 years of age.
Side effects
Major & minor side effects for Petril Plus (10/0.5 mg) Tablet
Confusion
Convulsions
Decreased urine output
Fast heartbeat
Constipation
Diarrhea
Dry mouth
Decreased appetite
Joint pain
Sleepiness
Nervousness
Uncontrolled body movements
Reduced intellectual abilities
Decreased sexual urge
Blurred vision
Muscle pain
Uses of Petril Plus (10/0.5 mg) Tablet
What is it prescribed for?
Comorbid Depression and Anxiety Disorder
This medicine is used for the treatment of patients suffering from both depression and anxiety disorders.
Related Product
Petril 0.5 Tablet
Petril Beta 10 Tablet
Petril Beta 20 Tablet
Petril Plus Tablet
Petril-MD 0.5 Tablet
When not to use?
Allergy
This medicine is not recommended for use in patients with a known allergy to escitalopram, other SSRIs, clonazepam, other benzodiazepines, or any other inactive ingredients present along with these medicines.
Monoamine oxidase inhibitors
This medicine is not recommended for use in patients undergoing treatment with Monoamine Oxidase Inhibitors (MAOIs) such as selegiline, moclobemide etc. There should be a time gap of at least 14 days between stopping treatment with MAOIs and beginning treatment with this medicine to avoid their interaction.
Pimozide
This medicine is not recommended for use in patients undergoing treatment with pimozide due to increased risk of heart rhythm disorders.
Liver disease
This medicine is not recommended for use in patients with an active liver disease due to the increased risk of worsening of the patient’s condition.
Acute angle Glaucoma
This medicine is not recommended for use in patients with a known history of acute angle glaucoma due to the increased risk of worsening of the patient’s condition.
Warnings
Warnings for special population
Pregnancy
This medicine is not recommended for use in pregnant women unless absolutely necessary. All the risks and benefits should be discussed with the doctor before taking this medicine.
Breast-feeding
This medicine is not recommended for use in breastfeeding women unless absolutely necessary. All the risks and benefits should be discussed with the doctor before taking this medicine. If the medicine is used, the infant should be monitored closely for any adverse effects.
General warnings
Pediatric population
This medicine is not recommended for use in patients below 18 years of age since the safety and efficacy of use are not clinically established.
Diabetes
This medicine should be used with caution in patients with a known history of diabetes due to the increased risk of altered blood glucose levels. Close monitoring of blood glucose levels is recommended for such patients. Appropriate dose adjustments may be necessary based on the clinical condition of the patient.
Abnormal bleeding
This medicine may increase the risk of abnormal bleeding in some patients. This risk is especially higher in patients with a history of bleeding disorders and patients undergoing treatment with aspirin, NSAIDs, anticoagulants, etc. Report any unusual bleeding or bruising, blood in the stool, etc. to the doctor immediately. Appropriate dose adjustments or replacement with a suitable alternative may be necessary based on the clinical condition of the patient.
Impaired kidney function
This medicine should be used with caution in patients with a known history of kidney diseases due to the increased risk of worsening of the patient’s condition. Close monitoring of kidney function is recommended for such patients. Appropriate dose adjustments or replacement with a suitable alternative may be necessary based on the clinical condition.
Suicidal tendencies
This medicine may cause suicidal behavior in some patients. This risk is especially higher on the prolonged usage of the medicine. Patients should be monitored closely for any changes in behavior and mood; any such changes should be reported to the doctor immediately. Appropriate dose adjustments or replacement with a suitable alternative may be required based on the clinical condition.
Interference with awareness and movements
This medicine may interfere with general awareness and body movements. It is advised that you do not perform any activities such as driving a vehicle or operating heavy machinery during treatment with this medicine.
Withdrawal symptoms
This medicine may cause withdrawal symptoms if the usage is stopped abruptly. This risk is especially higher after a prolonged usage of this medicine. The treatment should be withdrawn gradually under the close supervision of your doctor. Do not stop the use of this medicine without consulting your doctor.
Drug abuse
This medicine should be used with caution in patients with a known history of drug abuse. Chances of dependency are very high, especially if the intended use is for a long duration of time. Patients should be monitored closely for any changes in mood and behavior and any drug-seeking behavior; any such changes should be reported to the doctor immediately.
Increased salivation
This medicine may cause an increase in salivation; caution is advised while administering this medicine to patients with difficulty in handling secretions. This risk is especially higher in patients with respiratory disorders. Report any respiratory disorders to the doctor before beginning treatment with this medicine.
Dosage
Missed Dose
The missed dose should be taken as soon as possible. It is advisable to skip the missed dose if it is already time for your next scheduled dose. Do not double your dose to make up for the missed one.
Overdose
Seek emergency medical treatment or contact the doctor in case of an overdose.
Interactions
All drugs interact differently for person to person. You should check all the possible interactions with your doctor before starting any medicine.
Interaction with Alcohol
Description
N/A
Instructions
Consumption of alcohol is not recommended during treatment with this medicine due to the increased risk of serious adverse effects. These side effects may include dizziness, drowsiness, difficulty concentrating, breathing difficulties, etc.
Interaction with Medicine
Cimetidine
Lithium
CYP2C19 Inhibitors
CYP450 inducers
Triptans
Disease interactions
Seizure disorders
This medicine should be used with extreme caution in patients with a known history of seizure disorders due to the increased risk of recurrent seizures. Treatment should be discontinued if the patient develops seizures or if there is an increase in the frequency of seizures in patients with a previous diagnosis of epilepsy. Replacement with a suitable alternative should be considered in such cases.
Closed-Angle Glaucoma
This medicine is not recommended for use in patients with a known history of Closed Angle Glaucoma due to the increased risk of worsening of the patient’s condition. Replacement with a suitable alternative should be considered under your doctor’s supervision.
Respiratory disorders
This medicine should be used with caution in patients with a known history of respiratory disorders since it may worsen the patient’s condition. Close monitoring of lung function is recommended for such patients. Replacement with a suitable alternative treatment may be required in some cases based on the clinical condition.
Food interactions
Information not available.
Lab interactions
Information not available.
This is not an exhaustive list of possible drug interactions. You should consult your doctor about all the possible interactions of the drugs you’re taking.
General Instructions
Take this medicine exactly as instructed by your doctor. Taking the right quantity for the right duration is very critical since this medicine may be habit-forming. Close monitoring of the patient for any changes in behavior or mood is recommended during treatment with this medicine. Consult the doctor if you experience any undesirable side effects. Do not stop the use of this medicine without consulting your doctor.




Reviews
There are no reviews yet.